> MIMIC THE CARDIAC MUSCLE CONTRACTION USING MICROPATTERNS
This application note describes how to plate Cardiomyocytes differentiated from human pluripotent stem cells (hPSC-CMs) on micropatterns and how to achieve better alignment and maturity to mimic the in vivo behavior of cardiomyocytes.
> Controlled 2D geometry
> Compatible with high resolution imaging and immunofluorescence
> Single cell analysis
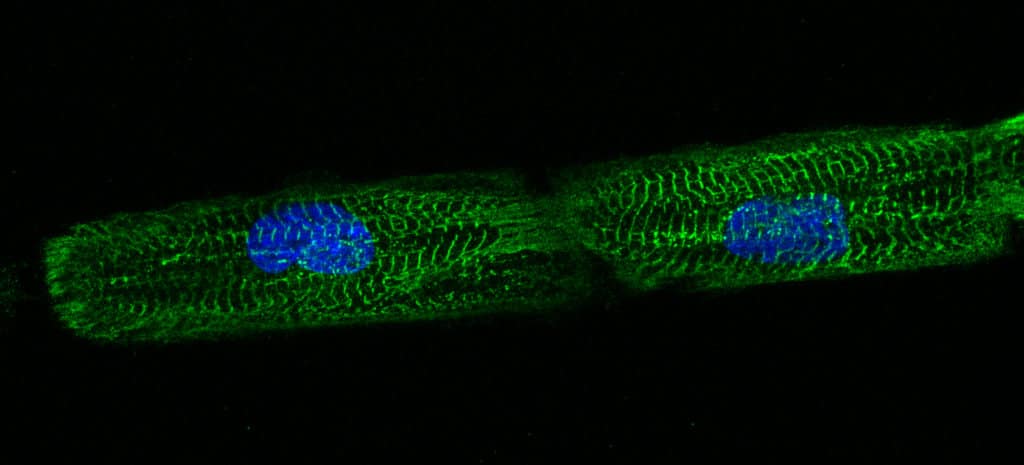
hPSC-CM on line micropatterns imaged by fluorescence microscopy.
Dussaud S., Jouve C., Hulot J.S., 2018.
PRINCIPLE AND BENEFITS
When hPSC-CMs are in a classic cell culture system, they organize themselves randomly on the substrate. By implementing standardized line-micropatterns, the goal is to force the cells to grow in a stripe configuration and study their individual behavior and as a whole. The purpose is to get closer to the real cardiac muscle cell configuration and contraction.
EXPERIMENTAL SETUP
> Material needed
Seeding substrate: Sterile micropatterned glass slides
Protein coating: fibronectin 50 µg/mL
Type of pattern: Lines with sizes from 10 to 100 µm
> Protocol
Protein coating
On a parafilm, pipette 100 µL of fibronectin
Gently place the micropatterned side of the slide on the drop
Incubate 1 hour
Gently rinse the slide with water and then PBS
Cell seeding
Purified hESC-CMs should be seeded onto micropatterns within 1 to 3 days after the purification step had been finished. This seeding step can be typically done between days 13 – 16 of overall cardiomyocyte culture.
Wash the purified hESC-CMs 3 times with PBS
Seed the cells at a density of 30 000 cells/cm² for confluent cell attachment
Line-patterned hESC-CMs can be compared with unpatterned cells on classic cell culture substrate.
You can also refer to the dedicated User Guide.
RESULTS
Results can be observed after a few days of culture (from 2 to 5 days is optimal). Samples must be treated for immunofluorescence imaging.
Unpatterned cells should have random shape and myofibrils directionality.
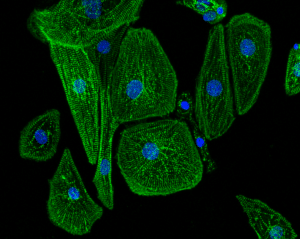
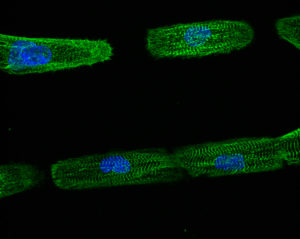
Images of unpatterned (left image) and line-micropatterned (right image) hPSC-CM.
Dussaud S., Jouve C., Hulot J.S., 2018.
Engineered hPSC-CMs have more physiological myofibril alignment, sarcomere lateral registry, cell shape, and direction of contractile movement than unpatterned hPSC-CMs (Ribeiro et al., 2015).

hPSC-CM imaged by fluorescence microscopy. This cell expresses Lifeact, which labels actin in myofibrils. The nucleus is labeled with Hoechst. Sarcomeres shorten along the pattern/cell major axis (Ribeiro et al., 2015).
On the micropatterned substrate, contraction should start occurring 24H after seeding the cells. Contractions among cells within a single feature are completely synchronized (Salick et al., 2014).
The seeded cells showed clear alignment with the features, dependent on the width rather than the overall aspect ratio of the features. It was determined that features with widths between 30 μm and 80 μm promoted highly aligned cardiomyocytes with a dramatic increase in sarcomere alignment relative to the long axis of the pattern (Salick et al., 2014).
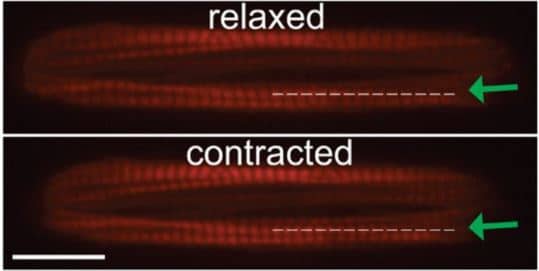
Sarcomere length during contraction and relaxation calculated in Lifeact-labeled cells from intensity profile of line scans (dotted line with green arrow) along myofibrils. (Scale bar, 20 μm.), Ribeiro et al., 2015.
CONCLUSION
Compared to classic cell culture substrates, micropatterned ones allow the complete control over the 2D geometry of the cells. In the particular example of hPSC-CMs, the cells showed clear alignment and directionality, as well as contractile synchronization among cells.
Line-micropatterns enable a much more relevant cell culture system for hPSC-CMs, to better reproduce the behavior of cardiac muscle cells.
REFERENCES
Micropattern Width Dependent Sarcomere Development in Human ESC-Derived Cardiomyocytes
Max R. Salick, Wendy C. Crone. et al. Biomaterials. 2014, May. 35(15): 4454-4464.
Contractility of single cardiomyocytes differentiated from pluripotent stem cells depends on physiological shape and substrate stiffness
Alexandre J. S. Ribeiro, Beth L. Pruitt et al. National Academy of Sciences.October 13, 2015.;112 (41) 12705-12710.
Dussaud S., Jouve C., Hulot J. S., 2018.
Experiments were conducted by Professor Hulot and his team using Cardiomyocytes differentiated from pluripotent stem cells on 4Dcell micropatterned glass slides. Results and images were nicely shared with 4Dcell.


