> CARDIAC ASSAYS & 3D CELL CULTURE [PART I]:
Cardiac spheroids & organoids
State of the art of innovative cardiac 3D cell culture systems
December 2023
Cardiac spheroids & organoids
State of the art of innovative cardiac 3D cell culture systems
December 2023
A necessary step in the path to adequately reproducing human tissue structure in vitro is the formation of complex 3D structures. Cells grown as monolayers on surfaces that are much stiffer than the human body are unlikely to behave similarly to their in vivo counterparts. One simple way to form 3D structures is to form spheroids or organoids of your cells of interest. Here we decipher the subtleties in the terminology, describe the current advances in the literature on cardiac organoids and spheroids, and discuss current techniques to create and culture spheroids and organoids.
> Spheroids vs. organoids: what’s the difference?
While the terms are occasionally used interchangeably, they do not mean exactly the same thing. They both designate self-assembled balls of cells. However, organoids can be considered to be a specialized type of spheroid. Typically, organoids arise from spheroids of stem cells in vitro that have been differentiated to many cell types and tissue layers that can be present in an adult organ[1].
In this manner, cardiac spheroids will generally be self-assembled from differentiated cardiomyocytes, with the possible addition of fibroblasts, endothelial cells, and smooth muscle cells to better imitate the cellular composition of the adult human heart. Cardiac organoids, on the other hand, will be formed and differentiated from pluripotent stem cells. The cellular composition of organoids will not be as well controlled as spheroids. However, the differentiation of the cells in situ, whether spontaneous or guided by chemical factors, will typically allow for a more complex structure to be formed, with a larger variety of cell types in various stages of differentiation.
> Cardiac organoids: what is the state of the art?
Cardiac organoids are balls of cells differentiated from one or several pluripotent stem cells. The mechanism by which these organoids are formed mimics the developments that occur during embryogenesis[2]. Cardiac organoids will typically be exposed to a cocktail of factors that will drive the cells through the stages of development of the heart, typically by modulating the WNT pathway. Using the GiWi (GSK-inhibition/Wnt-inhibition) protocol it is possible to generate cardiac organoids resembling crucial stages of early cardiomyogenesis, such as mesoderm specification, cardiac mesoderm induction, and cardiac cavity formation[2]. Cardiac organoids generated in this manner contain diverse cell subtypes, such as cardiomyocytes, endothelial, endocardial, smooth muscle, and epicardial cells, including endodermal derivatives[2].
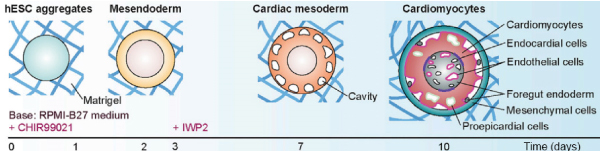
Figure 1: Stages of differentiation of cardiac organoids[3] using a protocol developed by Drakhlis et al.[4] Figure adapted from Kim et al.[3]
More recent differentiation protocols have allowed the formation of multi-chambered cardiac organoids (termed cardioids)[5]. This advance enabled the reproducible generation of cardiac organoids with compartment-specific in vivo-like gene expression profiles, morphologies, and functions. Notably, regions of the cardioids resemble the atria, left ventricle and right ventricle. These impressive feats of cell biology should have incredible potential to address diseases that affect the formation of heart chambers, interactions between heart chambers, and region-specific cardiac deformations during embryogenesis.
> Cardiac spheroids: what is the state of the art?
Unlike cardiac organoids, the cellular makeup of cardiac spheroids is determined by the terminally-differentiated cells used to create the spheroid. The exact cell types to include in cardiac spheroids is still up for debate. It is generally accepted that all of the major cells types present in the heart should be included. However, the heart is a heterogeneous organ, with different zones being composed of slightly different cell types. In addition, while the role of cardiomyocytes is evident, the role of other cells in the heart is less clear. Cardiac fibroblasts, for example, almost certainly play a structural role, for example depositing cardiac extracellular matrix. Recent reports have pointed towards roles for endothelial cells that exceed their function as vessel-lining cells[6]. It is likely that the exact cell types to use and their proportions will have to be adapted for the region of the heart that researchers are interested in. Nevertheless, several studies have already been carried out to determine the effect of inclusion of cell types other than cardiomyocytes on the phenotype of cardiac spheroids.
Cardiac spheroids that include hiPSC-derived endothelial cells, smooth muscle cells, and cardiac fibroblasts showed increased viability, sarcomere maturation and cardiomyocyte energy[7]. Spheroids composed of all four cell types also show greater field-potential duration and conduction velocity.
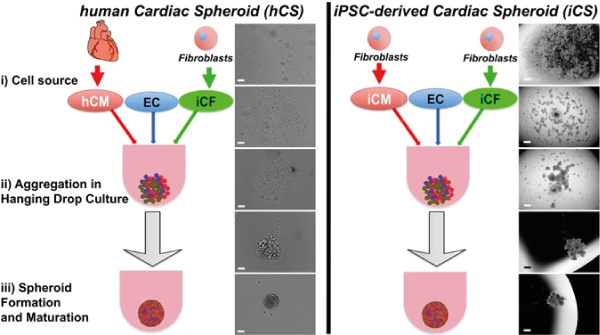
Figure 2: Formation of cardiac spheroids from human primary cells (left) and cells differentiated from iPSCs (right). Figure adapted from Polonchuk et al.[8]
Spheroids of cardiac cells can recapitulate the cellular organisation, extracellular matrix and microvascular network of human heart tissue[8]. In a study comparing spheroids created from adult human cardiomyocytes and iPSC-derived cardiomyocytes (co-cultured with primary endothelial cells and iPSC-derived cardiac fibroblasts), Polonchuk et al show indications of the formation of a micro-vascularized network lined with endothelial cells.
The choice of the cells to be used and the cocktail of factors that the cardiac spheroids are exposed to can also be chosen to create in vitro models of specific diseases. By treating their spheroids with TGF-beta, a known profibrotic agent, researchers created cardiac spheroids made of neonatal rat heart cells for fibrosis research[9].
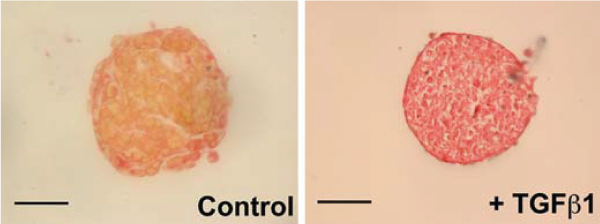
Figure 3: Staining of the muscular (yellow) and ECM (red) components of cardiac spheroids. The predominant red stain with the profibrotic agent TGF-beta demonstrates the creation of a fibrosis disease model. Scale bar, 100 μm.
Figure adapted from Figtree et al.[9]
> Spheroids and organoids: how are they created and how are they cultured?
Many techniques exist to culture spheroids, each with their advantages and their limitations[10]. The very basic necessary condition is that the cells must be prevented from adhering to surfaces, to prevent their spreading into a monolayer. This can be achieved by culturing the cells in vessels with non-adherent surfaces, or by keeping the cells in suspension so that they do not come into contact with surfaces. Examples of both of these are included in figure 4. However, the major disadvantage of these techniques is that the medium of the cells needs to be replenished, and in certain cases the medium needs to be spiked with drugs. Typical handling techniques (aspiration followed by pipetting new solution) have to be used with great caution as the spheroids are at risk of being lost.
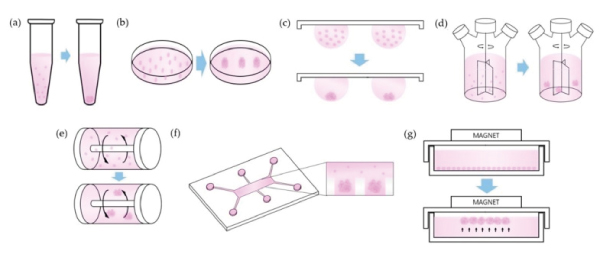
Figure 4: Techniques used to keep cell aggregates in suspension for spheroid tissue culture. Figure adapted from Ryu et al.[10]
Impressive results have also been achieved in hybrid formats wherein the attachment of cells is confined in 2 dimensions, resulting in a “2.5D” structure. In one publication, the authors used human iPSCs micropatterned in disk-shapes to form beating human cardiac microchambers[11] confined by the pattern geometry. Cell differentiation depended on the position of the cells in the micropattern, with an annulus of cells at the periphery expressing OCT4, indicating loss of pluripotency at the center of the micropattern.
While the authors prepared their own micropatterned surfaces, commercial solutions are readily available (4Dcell oxidation-resistant micropatterns).
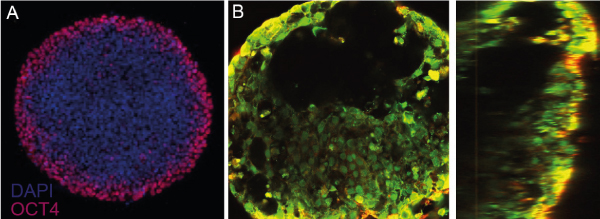
Figure 5: Micropatterned organoids. A) Loss of OCT4 expression at the periphery of the micropattern indicates loss of pluripotency. (Disk-shaped micropattern 600 um in diameter.) B) Following differentiation protocols, microchambers devoid of cells are visible in top (left) and side (right) views. Figure adapted from Ma et al.[11]
More recent advanced platforms allow the formation of multiple uniform spheroids per well in a multiwell plate format, suitable for high content screening, such as safety or toxicity studies. More advanced plates developed by 4Dcell present a contact point at the bottom of the microwells, allowing the spheroids to anchor in place while retaining their 3D format. This contact point allows media changes without perturbing the cells, or risk of losing them. The spheroid is surrounded by a hydrogel of tunable stiffness, imitating the mechanical properties of tissues in the body. In addition, the uniform size distribution allows studies of many near-identical spheroids or organoids at the same time. This homogeneity in size is particularly important for organoids, as the size of the initial spheroid of stem cells can dictate the subsequent differentiation of the cells. For more information about these spheroid plates, check out 4Dcell SmartSphero plates.
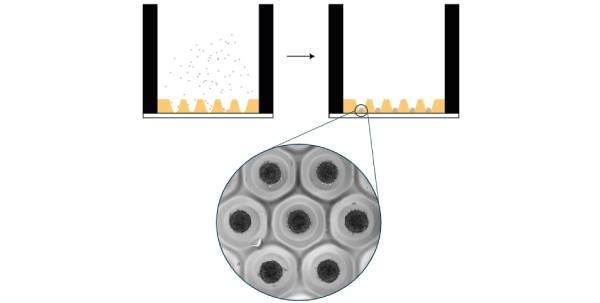
Figure 6: 4Dcell SmartSphero plates create homogenous spheroids, anchored at a small (~100 um) contact point, that are not lost during medium exchanges
> Conclusion
Spheroids and organoids have been a large part of the ongoing revolution in cell culture techniques that has seen researchers recognize the limitations of 2D cell culture and embrace 2.5D and 3D cell culture. While spheroids and organoids are an important step forward in understanding the relationships between cells and tissues, and understanding steps during embryogenesis, these 3D tissues still lack many of the important readouts that are necessary for cardiac tissue studies, in particular for toxicology and efficacy studies. Indeed, it is difficult to measure the action potential or contractility of cells in suspension. However, more advanced cell culture platforms, for example the SmartHeart developed by 4Dcell (which allows measurements of contractile forces of cardiac tissues in a multiwell plate format) are emerging. These will be the focus of an article to appear here soon! In the meantime, learn more about the SmartHeart.
> References